What do a spacecraft, a breathalyzer, and carbon monoxide have in common? Nothing at all – you’d think. And you’d be wrong! All three give you information on things that you cannot directly see, touch or measure. A spacecraft can capture some signal and send you beautiful images of a planet. With the help of a breath tester, a policeman may deduce the alcohol content in your blood. And using carbon monoxide, researchers may find highly reactive centers on materials surfaces. Let’s focus on the latter and see how it works!

When a molecule comes in contact with surface atoms, its properties change. By measuring these changes, you get information on the surface sites interacting with the molecule. Molecular vibrations – that you can measure by infrared spectra – provide very useful information: the vibration of carbon monoxide is very sensitive to the type of surface sites. That’s why this molecule is used to identify active centers on catalytic materials, such as titanium dioxide.
How does carbon monoxide (CO) bind to surface atoms? If you’re a chemistry student, you (should) know very well how CO interacts with molecules and ions. You’ve learned that this molecule can work both as a donor and as an acceptor of electron density. Well, what’s nice, is that this happens also on surfaces, and you can see it experimentally.
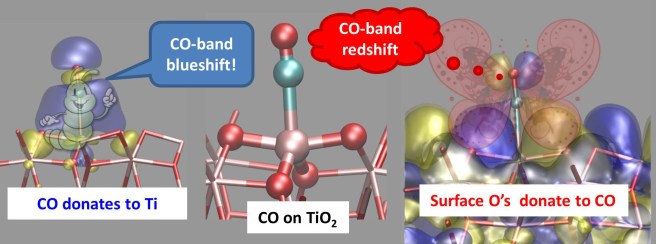
Let’s see this step-by-step. Carbon monoxide is a peculiar molecule. When it acts as a donor, charge flows to its bonding partner, which could be, for example, a metal cation. This process strengthens the C−O bond and increases its vibration frequency. This means that, in the infrared spectrum of the sample, you’ll find the CO band at higher frequencies – “blue-shifted” – with respect to the free, unperturbed molecule. But carbon monoxide can also accept electron density from its bonding partner. If this occurs, the C-O bond becomes weaker: its stretching frequency decreases, and you’ll see a “red-shifted” CO band in your spectrum.
Carbon monoxide is colorless, odorless, and highly toxic – a true and unmerciful silent killer. It binds to iron(II) in hemoglobin, and this prevents the delivery of oxygen to the human tissues. This is a – very unfortunate – case where carbon monoxide acts at the same time as a donor and as acceptor. The bond is synergic: CO donates to the metal, the metal back-donates to CO, and these two mechanisms reinforce each other:– that’s why it kills. This synergy occurs in many molecular complexes of transition metals and ions – often with less dangerous consequences. It’s less known on metal oxide surfaces, but it may happen as well. Is this the case of TiO2?
Not apparently, because carbon monoxide can only be a donor towards Ti cations – they are Lewis acids, and cannot give back electron density. The lower is their coordination number, the stronger is their acid power. For example, a Ti cation coordinated by 4 oxygens – Ti(4) – should be a stronger acid than one bound to 5 oxygens -Ti(5).
Researchers use carbon monoxide to explore the activity of surface cations and deduce their environment, in particular the number of oxygen neighbors. This information connects the reactivity of a catalytic center to its molecular structure, and may help them to improve the catalyst. Practically speaking, they send carbon monoxide on a TiO2 sample and measure the infrared spectrum. The rule is simple: the higher the frequency of the CO band, the more reactive are the Ti sites on the sample, and the lower their coordination number.
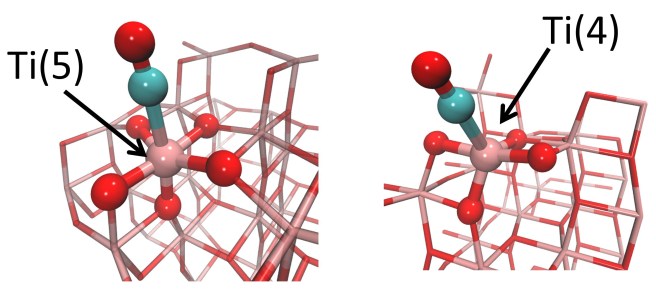
So if you had a TiO2 sample with Ti(5) sites, and a second one with Ti(4), what would you get from the experiment? “The second sample should show a more blue-shifted CO band, because Ti(4) is a stronger Lewis acid”. If you answered this, you’d be wrong… because we actually did the experiment, checked with calculations, and found the contrary. We found that CO on Ti(4) gives a less blue-shifted band – even if Ti(4) is a stronger Lewis acid. Just as if a breathalizer estimated a lower alcohol content in a drunker driver. This could happen only if a sort of magic potion neutralized the effects of alcohol (something similar exist in real life, but it’s a mineral and belongs to the large family of zeolites). Similarly, our carbon monoxide on Ti(4) should have received an antidote against the loss of electron density. The antidote could only be electron density: but where did it come from? Simply from the oxygen atoms bound to Ti(4): they are close enough to CO and ready to help.
In short, what happens is that CO donates electron density to Ti, but the surface oxygens donate electron density to CO. The first process strenghtens the C-O bond, but the latter has opposite effects. As a result, you find the CO signal at frequencies lower than expected. The two mechanisms are sketched in the figure below – my attempt to explain in a simple way the two-fold nature of the Ti-CO bond on titanium dioxide surfaces.
So, if you see high frequency bands in an infrared spectra of CO, please be warned: not necessarily they are due to very reactive sites on TiO2 surfaces. And also keep in mind that carbon monoxide gives you indirect information on your sample. Its signal can be influenced in complex ways by several factors – you might misinterpret your data, based on simple rules. From a practical viewpoint, i think that you should be aware of this, especially if you’re working on CO, or titanium dioxide materials. More speculatively, this story might help us to better understand how molecules interact with surface atoms. The complex, delicate balance of molecular-scale interactions is at the origin of technologically important phenomena – reactivity, catalysis, photocatalysis, just to mention some of them. Understanding these interactions more deeply could help us to improve their practical applications. Much effort is still needed, but it’s worth doing!
This research by our group has been published recently (Deiana et.al., ChemPhysChem 2016, 17, 1956; 10.1002/cphc.201600284). It was also sketched in a short summary, and by an infographics in a previous post. Here i used other words to tell the same story, because i feel it’s important to make research results accessible to a larger community.


